|
“Valence, Oxidation Number, and Formal Charge: Three Related but Fundamentally Different Concepts”. “Comprehensive definition of oxidation state (IUPAC Recommendations 2016)”. We write the charges of the atoms as H + and Cl –. From the oxidation state, you know the charge (or vice versa). But, hydrogen has an oxidation state of +1, while chloride has an oxidation state of -1. One electron from each atom participates in chemical bond formation. For example, in HCl, both H and Cl have a valence of 1. Valence: Valence is the number of electrons used by an atom to form a chemical bond.Ĭonfused? Usually, what you want to know is the oxidation state, which carries both a number and a positive or negative sign.In this case, the more electronegative atom gets the electrons. Oxidation State (Oxidation Number): Oxidation state is the charge on an atom when all of its ligands are removed heterolytically.In homolytic cleavage, electrons sharing a bond are split equally between the two atoms. Charge (Formal Charge): Charge is the electrical charge of an atom when all of its ligands are removed homolytically.These three terms are related, but have slightly different definitions: In many texts, the terms charge (or formal charge), valence, and oxidation number are used interchangeably. 
When we talk about the charge on an atom, it’s usually its oxidation number rather than its valence. Table of common element charges All element valence charges for hydrogen through rhodium. The name of each element (in brown) is accompanied by its chemical symbol (in red), Group I a. 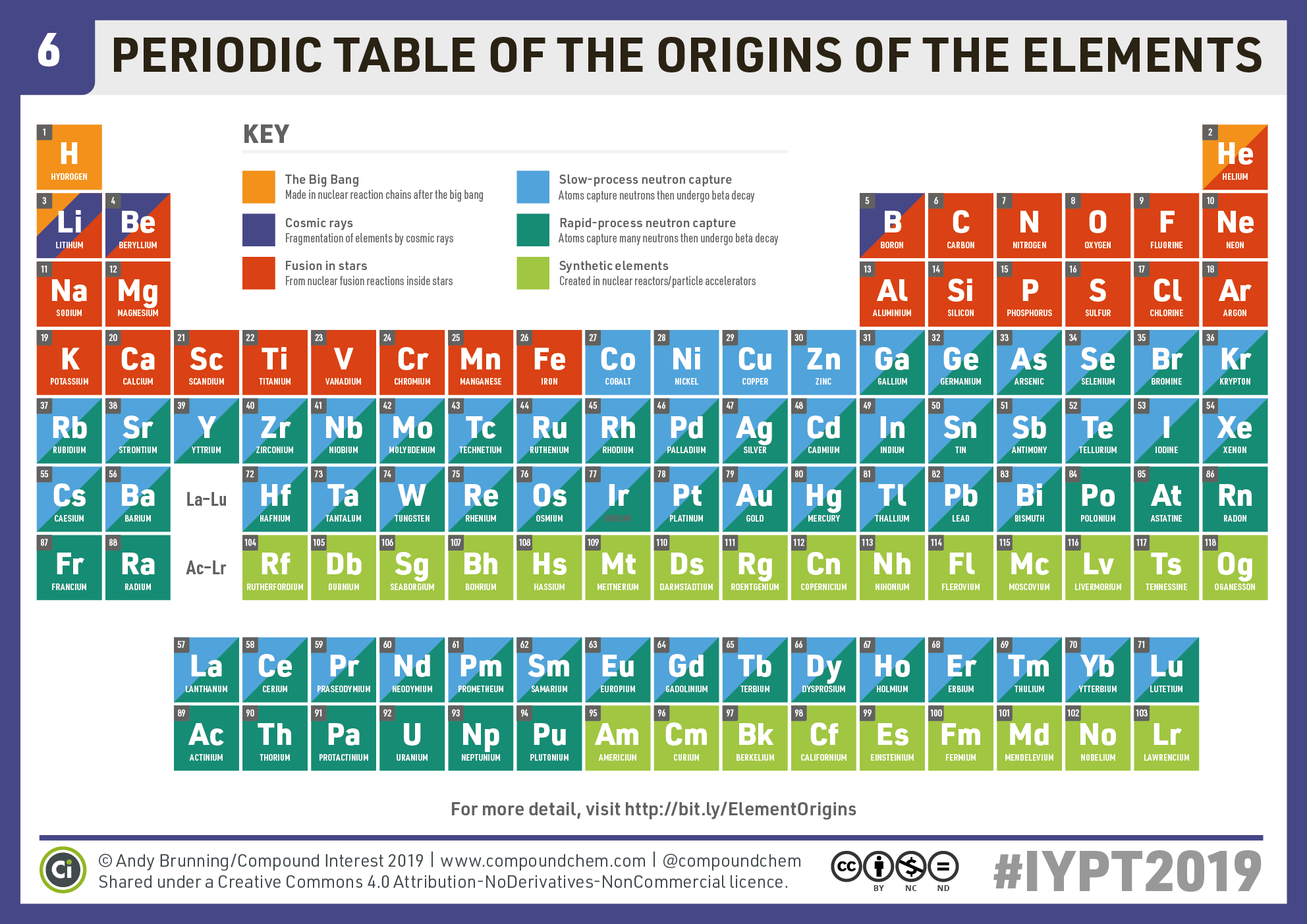
Find the charge by balancing charge in a compound. ELEMENTS IN SAME COLUMN (GROUP) HAVE SIMILAR CHEMICAL PROPERTIES.For a single atom, the charge is the number of protons minus the number of electrons.The first shows common element charges, while the second shows all the element charges for the first 45 elements (most common charges in bold). 
Charts come from empirical data on the real behavior of elements, which may differ somewhere from the periodic table predictions. Lanthanide and Actinide Series: Variable positive charges Groups 3-12 (Transition Metals): Variable positive charges It is a graphic formulation of the periodic law, which states that the properties of the chemical elements exhibit a periodic dependence on their atomic numbers. It is widely used in chemistry, physics, and other sciences, and is generally seen as an icon of chemistry. The usual charge of an element is common to its group. The periodic table, also known as the periodic table of the ( chemical) elements, is a tabular display of the chemical elements. There are four ways to find the charge of an element: You can download and print these graphics and tables for references. The difference between charge, oxidation state, and valence is explained, too. Here is a chart of element charges and an explanation of how to find the charge of an element if you don’t know it. This entry was posted on Septemby Anne Helmenstine (updated on February 14, 2022)Ītoms of the elements display a range of charges, but you can predict the most common charge of most elements using its element group.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
